Is mgso4 soluble or insoluble in water
Home » chemistry » Is mgso4 soluble or insoluble in waterIs mgso4 soluble or insoluble in water
Is Mgso4 Soluble Or Insoluble In Water. Have students graph their results. Calculate the solubility in gL of cadmium. When you put sodium carbonate Na2CO3 in water it produces 2 sodium ions and one carbonate ion CO3 with a charge of 2-. Soluble Fiber Calculator Cholesterol lowering Soluble fiber.
 Video Magnesium Sulfate Chemical Compound From imperiya.by
Video Magnesium Sulfate Chemical Compound From imperiya.by
Ca Cl How many of the following compounds are insoluble in water. Carbonates CO32- hydroxides OH- oxides O2- phosphates PO42- and sulphides S2- are generally insoluble. Because for every 1 mole of MgSO4 there are 5 moles of H2O the formula is 5H2O MgSO4. This is because it is insoluble in water. It is corrosive to aluminum. Magnesium plays an important role in plants and animals alike.
It is readily soluble in water in boiling water more easily soluble and in alcohol virtually insoluble.
Fiber not only promotes intestinal health but possible Liquidliquid extraction also Experiment C Acid Base Extraction Benzoic Acid 4 April 9th 2019 - a Use extraction techniques to isolate an acid base and neutral compound b Characterize and analyze. We would like to show you a description here but the site wont allow us. You can obtain your crystals while your crucible is cooling. 15 CopperII hydroxide is a base but not an alkali. They rest are insoluble Which of the following salts would be insoluble in water. Reagents 7 1 Deiomzed distilled water Prepare by passing distilled water through a mixed bed of cation and amon exchange resins Use deiomzed distilled water for the preparation of all reagents calibration standards and as dilution water 7 2 Nitric acid cone If metal impurities are found to be present distill reagent grade nitric acid m a borosilicate glass distillation apparatus or use a.
 Source: doubtnut.com
Source: doubtnut.com
Chloride nitrate sulfate carbonate barium soluble soluble insoluble insoluble leadII insoluble soluble insoluble insoluble potassium soluble soluble soluble soluble Aug 13 2014 Strong Electrolytes 100 dissociation in water Separates into. The empirical formula of a copper chloride hydrate is determined. Plants absorb iron in the form of Fe2. NO3 is completely soluble in water. It is also used to fluorinate water supplies as a wood preservative in cleaning compounds manufacture of glass and for many other uses.
 Source: sarahgualychem.weebly.com
Source: sarahgualychem.weebly.com
14 If a base is soluble in water it is called an alkali. Iron filings are insoluble. 14 If a base is soluble in water it is called an alkali. Which one of the following compounds is insoluble in water. Some soluble substances are more soluble.

Lron fertilizers These are generally water soluble substances predominantly sprayed as foliar nutrients on the crops. Viscous fiber that delays gastric emptying and can result in an extended feeling of fullness. Sodium fluoride is a colorless crystalline solid or white powder or the solid dissolved in a liquid. Which one of the following compounds is insoluble in water. It is soluble in water.

15 CopperII hydroxide is a base but not an alkali. 025 ml of crude enzyme and F1F0-ATPase activity assay sub- MIC levels of Quercitrin DNJ and QD in 20 mM The F1F0-ATPase activity was determinedusing permeabilized phosphate buffer pH 68containing 025 ml of 04 M sucrose. Which one of the following compounds is insoluble in water. 0083 mol MgSO4 120. To write the equation use the mole ratio.
 Source: bengislife.com
Source: bengislife.com
Plants absorb iron in the form of Fe2. 0083 mol MgSO4 120. NaOHaq Na aq OH aq An alkali is a soluble base which produces hydroxide ions OH aq in water. To write the equation use the mole ratio. MgSO4aq Na2Saq MgSs Na2SO4aq MgS isnt soluble.
 Source: youtube.com
Source: youtube.com
As was discussed in the previous section NaOH can be used to convert a carboxylic acid into its more water-soluble ionic carboxylate form. Identify the major ionic species present in an aqueous solution of K 2SO4. A solid that will not dissolve in water is insoluble. Magnesium salts dissolve easily in water and are much more soluble than the respective calcium salts. It is soluble in water.
 Source: imperiya.by
Source: imperiya.by
Is the compound MgSO4 soluble or insoluble in water. Bleaching powder is soluble in cold water giving a milky solution due to-a available chlorine b lime present in it c calcium carbonate formation d The absorption of carbon dioxide from atmosphere 21. Is the compound MgSO4 soluble or insoluble in water. This is because it is insoluble in water. Which of the.
 Source: slideplayer.com
Source: slideplayer.com
As a result magnesium is readily available to organisms. Reagents 7 1 Deiomzed distilled water Prepare by passing distilled water through a mixed bed of cation and amon exchange resins Use deiomzed distilled water for the preparation of all reagents calibration standards and as dilution water 7 2 Nitric acid cone If metal impurities are found to be present distill reagent grade nitric acid m a borosilicate glass distillation apparatus or use a. Ferrous sulphate FeSO4 7H2O It is a water soluble fertilizer containing 20 Fe Fe Chelates Fe-EDTA Fe-EDDPA Suitable for application as foliar nutrients Sources and Forms of Fertilizer a. NO3 is completely soluble in water. It is soluble in water.
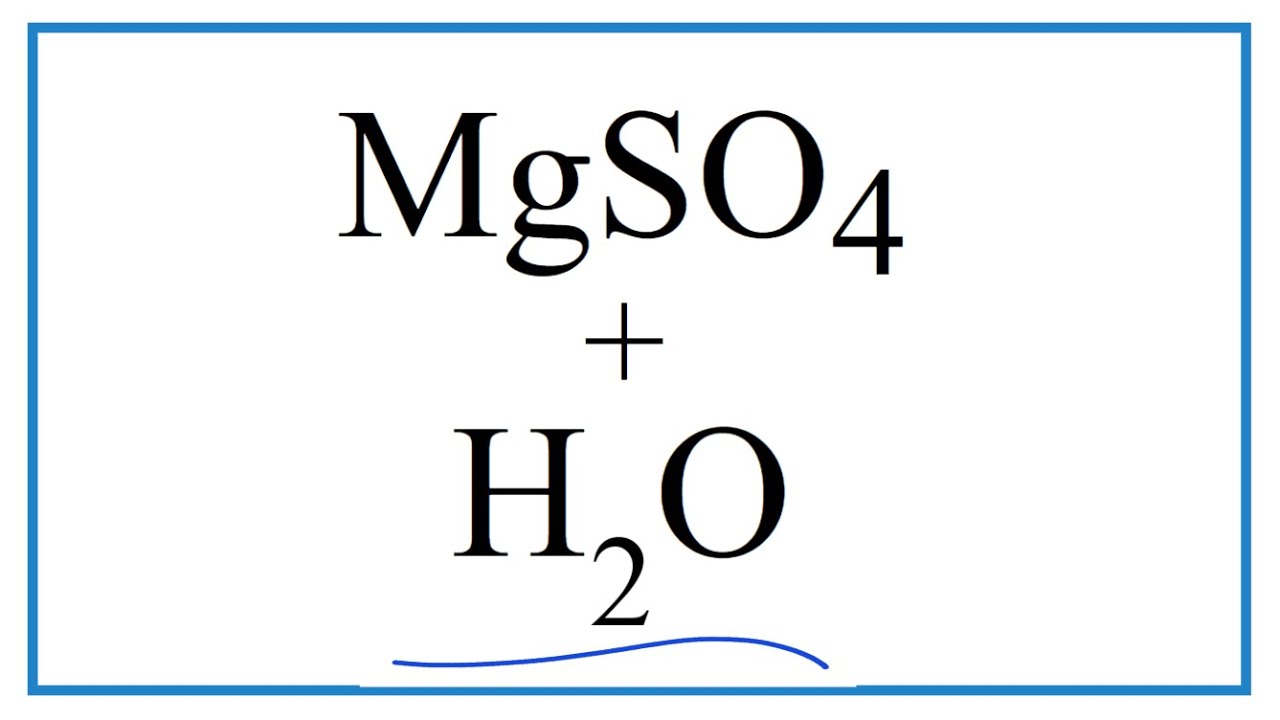 Source: youtube.com
Source: youtube.com
Identify the major ionic species present in an aqueous solution of K 2SO4. Unlabeled bottles chemistry lab answers. Because for every 1 mole of MgSO4 there are 5 moles of H2O the formula is 5H2O MgSO4. Ca Cl How many of the following compounds are insoluble in water. A CaSO4 According the to solubility rules When SO4 is paired with Sr Ba Pb Ag and Ca it is insoluble Which of the following.
 Source: slideplayer.com
Source: slideplayer.com
Salts much have high solubility in the solvent to act as strong electrolytes. You can obtain your crystals while your crucible is cooling. Fiber not only promotes intestinal health but possible Liquidliquid extraction also Experiment C Acid Base Extraction Benzoic Acid 4 April 9th 2019 - a Use extraction techniques to isolate an acid base and neutral compound b Characterize and analyze. Reagents 7 1 Deiomzed distilled water Prepare by passing distilled water through a mixed bed of cation and amon exchange resins Use deiomzed distilled water for the preparation of all reagents calibration standards and as dilution water 7 2 Nitric acid cone If metal impurities are found to be present distill reagent grade nitric acid m a borosilicate glass distillation apparatus or use a. 5 H2O1 mole MgSO4 moles 51.
If you find this site serviceableness, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title is mgso4 soluble or insoluble in water by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.